INNOVATION • EDUCATION • SUPPORT
The Total Knee Replacement
TKR is a surgical procedure in which damaged joint surfaces are replaced with a prosthesis designed to restore pain-free knee function. The replacement joint consists of a metal component attached to the end of the femur and a polyethylene component attached to the tibia. The components create new bearing surfaces that mimic normal movement of the joint.
It is the only treatment that fully restores life-long mobility to patients with acute osteoarthritis of the knee.
KNEE REPLACEMENT SOLUTIONS FOR EVEN THE LARGEST OR SMALLEST BREEDS
BIOMEDTRIX TOTAL KNEE REPLACEMENTS COMPLETED WORLDWIDE SINCE 2005
OF ALL BIOMEDTRIX TKR RECIPIENTS ARE EXPECTED TO RECOVER FULL MOBILITY
PATELLOFEMORAL OSTEOARTHRITIS
The primary goal of TKR surgery is to alleviate the pain and/or disability associated with knee arthritis and restore pain-free function. In most cases, it is possible to restore the joint function to a level similar to that of a healthy knee joint, and long-term medical management is not required. In addition to significant cost savings, this will eliminate the potential problem of long-term complications associated with chronic use of anti-inflammatory drugs.
To determine if TKR is the appropriate option, schedule a consultation with your veterinarian. This is a surgical procedure but, for the right patient, surgery can greatly improve quality of life.
Recipients of BioMedtrix Total Knee Replacement should be able to use the new joint for the rest of their lives. Patients are expected to recover full mobility, and suffer no lameness or muscle atrophy, overcompensation, or limitations of any kind.
SURGICAL OVERVIEW
A TKR procedure consists of the removal and replacement of the entire knee joint. During surgery, the knee joint is exposed through an incision on the side of the knee. The joint surfaces are replaced, stability of the knee ensured, and the surgeon verifies that the knee moves smoothly through a full range of motion. A light dressing is applied over the incision in the early post-operative period.
The Total Knee Replacement requires special training to be performed reliably. Surgeons who perform the procedure have been given extensive training and resources to ensure the best possible outcome for dogs, with the expectation of return to full mobility.
Originally adapted from the analogous procedure in human orthopedics, this procedure continually benefits from the expertise and support of pioneers in the veterinary industry.


RECOVERY
Activity is limited for two to six weeks following surgery. Dogs usually use the leg the evening of surgery, and most are doing very well in six months.
Implant survival depends on long-term stability of the implant, maintenance of a smooth articulation between the components, and the patient activity level. We expect that the implants will survive for the lifetime of a dog.

BioMedtrix has incorporated successful design concepts from human knee reconstruction into our Total Knee System and complemented them with specific design features based on the canine anatomy and biomechanics. The system focuses on accuracy and alignment of the preparation for restoration of the knee biomechanics, and an extensive array of implant sizes which are completely interchangeable allowing for optimum patient fit.
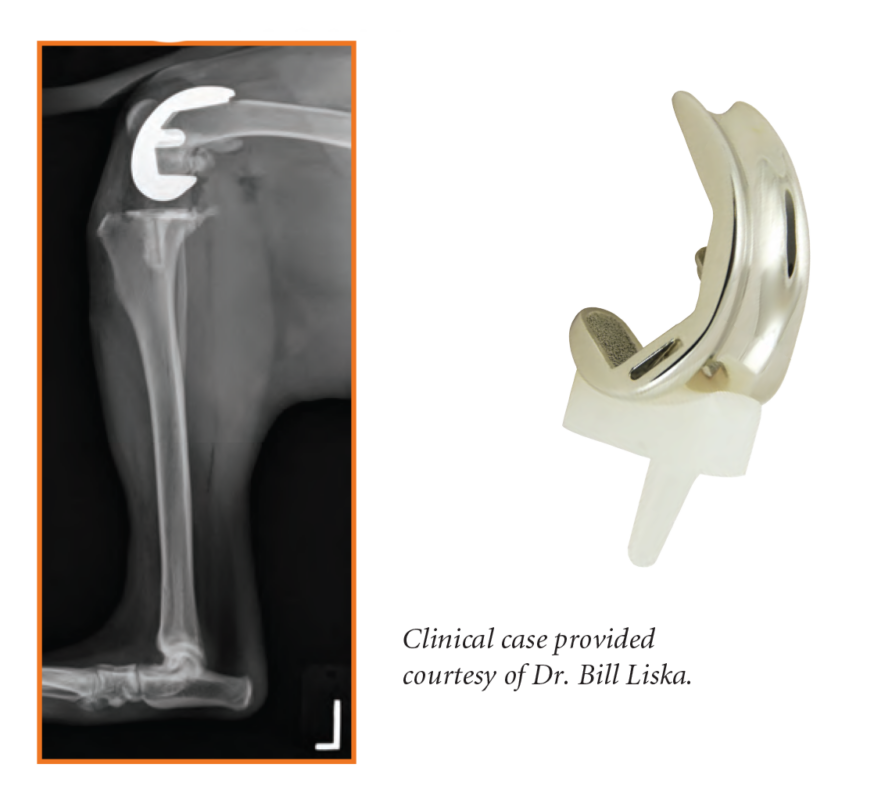
The implant system consists of seven (7) symmetrical femoral components (left or right) and five (5) symmetrical tibial components in thicknesses of 5mm, 7mm and 9mm in each size. Femoral components have a porous ingrowth surface for biologic fixation. Tibial components are available in all-polyethylene for cement fixation, and an optional porous-metal tray and polyethylene insert design for biologic fixation.
Complete interchangeability between femoral sizes and tibial sizes helps to optimize patient fit in a wide range of dog sizes and breeds.
FREQUENTLY ASKED QUESTIONS
TKR is a surgical procedure in which damaged joint surfaces are replaced with a prosthesis designed to restore pain-free knee function.
The replacement joint consists of a metal component attached to the end of the femur and a polyethylene component attached to the tibia. The components create new bearing surfaces that mimic normal movement of the joint.
TKR is recommended because it is an option for alleviating the pain and/or disability associated with knee arthritis. Many dogs that do not respond to standard medical or surgical management can benefit from TKR.
To determine if TKR is the appropriate option, schedule a consultation with your veterinarian. This is a surgical procedure but, for the right patient, surgery can greatly improve quality of life.
The knee joint is exposed through an incision on the side of the knee. The joint surfaces are replaced, stability of the knee ensured, and the surgeon verifies that the knee moves smoothly through a full range of motion. A light dressing is applied over the incision in the early post-operative period.
The primary goal of TKR surgery is to restore pain-free function. In most cases, it is possible to restore the joint function to a level similar to that of a healthy knee joint. Dogs that have had successful TKR surgery do not require long-term medical management. In addition to significant cost savings, this will eliminate the potential problem of long-term complications associated with chronic use of anti-inflammatory drugs.
The most significant risks associated with TKR surgery are anesthesia, implant loosening, and infection. Infection is a rare but potentially devastating complication of any surgical procedure. The risk of loosening is also rare. The risks associated with anesthesia are very low as a result of preoperative screening, modern drug use protocols, and intraoperative monitoring. Your veterinary team will do everything possible to minimize the risks.
If your dog’s arthritis is effectively controlled with drug therapy or other methods, TKR surgery is not likely to be recommended. It is important to recognize that TKR may be indicated if medical therapies eventually become ineffective.
Pre-existing infection is a contraindication to TKR surgery. Additional reasons why surgery may not be recommended include lameness associated with arthritis in other joints, neurological disease, and previous amputation of a forelimb or opposite hind limb. These conditions do not necessarily preclude TKR but the final decision is made on a case-by-case basis.
The canine TKR system was introduced in 2005. Since that time, over 300 surgeries have been preformed throughout Australia, Europe, the United States, and Canada. We anticipate demand for this surgery will increase significantly each year.
Available data indicates that TKR is effective in providing pain-free function in dogs with end-stage arthritis. As part of the ongoing clinical trial of canine TKR, BioMedtrix is actively collecting clinical data. The results from this prospective study will provide a clear picture regarding the overall long-term success rate for TKR in dogs. Initial results of the clinical trials have been published. It is our expectation that success rates will be similar to those seen with total hip replacement (THR) in dogs which are widely cited as being successful.
There is not enough data beyond 5 years of follow-up to provide a definitive answer to this questions. In humans, TKR implants are expected to last more than fifteen years. Implant survival depends on long-term stability of the implant, maintenance of a smooth articulation between the components, and the patient activity level. We expect that the implants will survive for the lifetime of a dog.
As with all surgical procedures, patient-owners should consult with their veterinary orthopedic surgeon prior to surgery concerning any potential risks associated with surgery.
BioMedtrix Total Joint Replacement implants are available in a wide range of sizes and configurations to address various anatomic morphologies, and are manufactured to precise tolerances to ensure optimal performance and long-term success. The use of BioMedtrix implants in a hybrid configuration with other implant systems has not been validated by BioMedtrix, and may result in implant failure. For specialty cases, custom implants may be designed and produced upon request.